Foodborne antioxidants from berries used in our PC products

Information on food-borne antioxidants, particularly those contained in the berries that make up Phytonutrient Canada’s products
1.0 Introduction
The purpose of this scientific section is to inform consumers about the scientific aspects that motivate the development and marketing of Phytonutrient Canada’s products and to briefly explain the very complex mechanism that allows foodborne antioxidants to play a very important role in our health. Particular emphasis will be placed on polyphenols, antioxidants found particularly in certain berries including wild blueberries and cranberries.
2.0 Broad definition of an antioxidant
An antioxidant is a molecule that decreases or prevents theoxidation of other chemicals. This reaction can produce free radicals that cause destructive chain reactions. Antioxidants are able to stop these chain reactions by reducing themselves with free radicals and thus annihilating their action.
3.0 History
The origin of the term antioxidant is not medical or medicinal. This word was originally used to refer to chemicals that prevent reactions with oxygen. It was towards the end of the nineteenth century and the beginning of the twentieth century that the properties of antioxidants were widely studied for their use in industrial processes in order to reduce, for example, the vulcanization of rubber and corrosion of metals.
In biology, it was only with the identification of vitamins A, C and E that the importance of antioxidants in the biochemistry of living organisms appeared.
4.0 Oxidative stress and its effects on health
Oxygen is essential for our survival, but its use by the body’s cells is not without danger. A significant portion (1-2%) of the oxygen we breathe is converted into toxic derivatives called free radicals (1). Free radicals can react and damage cellular components like proteins, lipids, and DNA (2). The oxidation of biomolecules by free radicals is involved in several pathologies such as cardiovascular diseases (arteriosclerosis), neurodegenerative diseases (Parkinson’s, Alzheimer’s), cancer as well as aging (3-7). Several enzymes and small molecules eliminate free radicals including vitamins E and C (8). Plants and animals use and produce many antioxidants to protect themselves, such as glutathione, vitamin C and vitamin E. For its part, humans are able to produce from the amino acid cysteine,a powerful antioxidant, alpha-lipoic acid, also called lipoate.
During aging, the generation of free radicals increases and their amount becomes greater than the capacity of antioxidant defenses. This imbalance, called oxidative stress, will lead the body to a pathological state (9). Consumption rich in antioxidant compounds can mitigate the impact of this oxidative stress. This imbalance can also be created by lifestyle habits such as smoking, lack of sleep, excessive alcohol consumption or poor diet.
5.0 Oxidant/Antioxidant
Antioxidants are biological substances found in food that have the role of preventing oxidation reactions caused by free radicals. By definition, it is a substance with a low concentration compared to that of the oxidisable substrate capable of delaying or stopping the oxidation of the substrate. Antioxidants are naturally present in plant foods in the form of substances such as flavonoids and carotenoids, minerals, and vitamins A, C, or E.
The best known antioxidants areß-carotene (provitamin A),ascorbic acid (vitamin C), tocopherol (vitamin E), polyphenols and lycopene. These include tannins (cocoa, coffee,tea, grapes,etc.), flavonoids (pear apple, honey, etc.), anthocyanins (wild blueberry, cranberry, lingonberry idaea, etc.) and phenolic acids (artichoke, rosemary, etc.).
In the scientific field, antioxidants have been classified into three groups, namely:
- vitamins
- minerals
- phytochemicals (organic chemical compounds that can be found in foods of plant origin)
Duringa balanced meal, these three groups of antioxidants are easily found.  As for dietary supplements, the majority of them contain synthetic antioxidants. On the other hand, it seems that antioxidants from food sources have a much higher bio activity than those from supplements from synthetic extracts. This is due on the one hand to the fact that foods contain greater heterogeneity of antioxidants and on the other hand that synthetic antioxidants are less well absorbed by the human body. Phytonutrient Canada’s liquid products are therefore easily assimilated by the body.
As for dietary supplements, the majority of them contain synthetic antioxidants. On the other hand, it seems that antioxidants from food sources have a much higher bio activity than those from supplements from synthetic extracts. This is due on the one hand to the fact that foods contain greater heterogeneity of antioxidants and on the other hand that synthetic antioxidants are less well absorbed by the human body. Phytonutrient Canada’s liquid products are therefore easily assimilated by the body.
Antioxidants fall into 4 categories:
- carotenoids
- vitamins
- trace elements
- polyphenols
- Classification of major classes of foodborne antioxidants
7.0 Polyphenols
Polyphenols are chemical compounds naturally present in the plant kingdom, especially in northern berries. There are more than 8000 different phenolic molecules, such as simple molecules such as phenolic acids, much more complicated molecules (tannins) and others that can be complexed to sugars, proteins and even lipids. Among the polyphenols, we find flavonoids which represent the main group of this series. Indeed, flavonoids are present throughout the plant kingdom, with the exception of fungi and algae. They are derivatives of water-soluble (water-soluble) polyphenols, often colorless or yellow (with exceptions, such as anthocyanins). Flavonoids are in themselves an extremely large family of compounds, playing important physiological roles (nutritional, medicinal, UV filters…).
They are particularly present in Quebec berries, are of particular interest to human health and are the subject of many medicinal claims, particularly for their strong antioxidant capacity.
7.1 Flavonoids
Flavonoids are themselves classified according to their degree of oxidation into subgroups such as:
– flavonols
– flavones
– flavanols
– isolflavones
– proanthocyanidins
– anthocyanins
7.2 Isoflavones
Isoflavones are compounds found mainly in legumes (for example in soybeans whose phytoestrogenic properties are widely studied). Some isoflavone derivatives are powerful agents against certain bacteria. They have bacteriostatic properties and are specifically induced during infections by plant pathogenic organisms. In particular, many phytoalexins are found in legumes (e.g.: phaseollin from beans, glyceolline from soybeans)
7.3 Proanthocyans
Proanthocyanins, also called proanthocyanidins, are flavonoid compounds present in many plants especially in the skin and grape seeds which explains their abundance in red wine.
7.4 Anthocyanins
Anthocyanins are flavonoids found in the plant kingdom that give the characteristic color of leaves, flowers and fruits. These are colored compounds (orange, purple to blue) and usually water-soluble. Unlike other flavonoids, anthocyanins absorb most of the time in the ultraviolet spectrum. They play a major part in the coloration of petals, but they are also found in many plant tissues. Their synthesis in the leaf organs is often activated by stress (cold, deficiencies, senescence …). Their compounds are very often used as food colorings and have very strong antioxidant properties.
Anthocyanins have been studied based on several biological activities including antioxidant capacity, effect on capillary vessel permeability and fragility, aggregation of blood platelets and effect on collagen (w-5). The antioxidant capacity of anthocyanins is one of the most important biological properties. Epidemiological and biomedical research suggests that antioxidants contained in berries such as blueberries, lingonberry and cranberries may play a preventive role in the onset of certain diseases such as cancer, cardiovascular diseases and neurodegenerative diseases such as Alzheimer’s and Parkinson’s. Anthocyanins and other flavonoids make a substantial contribution to the total antioxidants of the diet (w6-7).
The anthocyanins contained in BMC products are said to be a natural antioxidant 20 times more effective than vitamin A and C.
8.0 The Role of Foodborne Antioxidants on Health
The majority of fruits, vegetables, spices or herbs that we consume have for the most part nutritional and organoleptic intakes important but some of them can also bring some specific health benefits. It is therefore very advantageous to also know their medicinal content.
Not all foodborne antioxidants are born equal and do not all do the same job. The antioxidant content is very different from one food to another. For a need for vitamin C, we suggest orange, kiwi and / or plaquebière. For vitamin E, it will be sea buckthorn and for a provitamin A intake, it will be carrot, apricot and / or spinach. To mitigate the risks of cardiovascular diseases and degenerative diseases, wild blueberries or lingonberry idaea should be thought of for their high concentration of anthocyanins. To help prevent recurrent urinary tract infections, Health Canada recognizes that at a certain minimum level of daily consumption, cranberry products help protect against them, which bananas and pears do not.
To date, only the role of a very small part of foodborne antioxidants is known. There are more than 8000 phenolic molecules, of which only a very small part has been the subject of extensive and recognized scientific studies. These antioxidant molecules do exist but their exact roles on humans remain unknown to this day.
9.0 Berries Consumption and Its Effects on Health
 We already knew the benefits of a balanced diet of fruits and vegetables for the maintenance of a healthy lifestyle. Canada’s Food Guide recommends consuming between 8 to 10 servings of fruits and vegetables per day. Indeed, consuming these recommended amounts reduces the risk of the appearance of several diseases. Quebec’s boreal forest, which has more than 850 species of vascular plants (10),represents an abundant source of compounds useful for the treatment and prevention of various diseases. Several plants in this vast ecosystem contain a high concentration of antioxidant compounds. Wild dwarf blueberries, lingonberry, plate bog and some other berries are renowned for their high content of antioxidant compounds and, therefore, are often considered references at this level. Numerous studies show that antioxidants found in boreal berries possess remarkable protective properties (10-58).
We already knew the benefits of a balanced diet of fruits and vegetables for the maintenance of a healthy lifestyle. Canada’s Food Guide recommends consuming between 8 to 10 servings of fruits and vegetables per day. Indeed, consuming these recommended amounts reduces the risk of the appearance of several diseases. Quebec’s boreal forest, which has more than 850 species of vascular plants (10),represents an abundant source of compounds useful for the treatment and prevention of various diseases. Several plants in this vast ecosystem contain a high concentration of antioxidant compounds. Wild dwarf blueberries, lingonberry, plate bog and some other berries are renowned for their high content of antioxidant compounds and, therefore, are often considered references at this level. Numerous studies show that antioxidants found in boreal berries possess remarkable protective properties (10-58).
Several studies have also shown that, for a healthy person, the consumption of fruits rich in antioxidants reduces the risk of developing cardiovascular disease, cancer, obesity and diabetes (w 2-3). Since the publication of these results, the phenolic compounds of fruits have been the subject of much work aimed at enhancing their nutritional and biological properties. In recent years, several techniques have been proposed to concentrate and purify antioxidants from fruits, and even to produce synthetic compounds from them highlighting that the consumption of fruits and berries allows the maintenance of a balance between pro-oxidants and antioxidants. Although berries are referred to as small only according to mass or volume criteria, studies show that these berries generally have much higher concentrations than other fruits with compounds of high nutritional value. These berries are of particular interest from a nutritional and medicinal point of view especially for their high concentration of antioxidants.
 Wild blueberries, cranberries and elderberry (cough syrup) are among, if not the only fruits that can be used as medicinal ingredients to develop consumable natural health products recognized by Health Canada. We are talking about years of scientific research often supported by ancestral medicinal knowledge before organisms such as Health Canada can recognize that a plant, or part of itself, has an effect on a specific function of the human organism and that this effect is measurable. It is only from this official acceptance, which involves in its process of accepting evidence of efficacy on humans, that a plant material can be used to develop natural health products or drugs approved by Health Canada.
Wild blueberries, cranberries and elderberry (cough syrup) are among, if not the only fruits that can be used as medicinal ingredients to develop consumable natural health products recognized by Health Canada. We are talking about years of scientific research often supported by ancestral medicinal knowledge before organisms such as Health Canada can recognize that a plant, or part of itself, has an effect on a specific function of the human organism and that this effect is measurable. It is only from this official acceptance, which involves in its process of accepting evidence of efficacy on humans, that a plant material can be used to develop natural health products or drugs approved by Health Canada.
Even if they are associated with exceptional health virtues by their manufacturers and distributors, exotic fruits such as Goji, acai and noni cannot be used for these purposes. Mangosteen is another example. It is true that some of them have high antioxidant potential but no good quality clinical trials confirming these claims have been published. We already have access in Canada to a wide variety of local berries and vegetables that have a high antioxidant value and clinically proven beneficial properties such as: blueberry,cranberry, strawberries, raspberry,carrot, tomatoes, broccoli, onion etc. The high price of these exotic whole juices or fruits and the lack of reliable clinical data on them therefore do not justify, at least at present, their addition to our diet.
10.0
- ORAC (OxygenRadical Absorbance Capacity) measurement
The antioxidant market has exploded and a multitude of products have appeared on the market. The arguments most often used by manufacturers to judge the effectiveness of their products were and often still remain, the ORAC value. The higher the ORAC, the better and more effective the product supposedly. This advance is not always true. Moreover, the ORAC value was so misused by the industry that the United States Department of Agriculture (USDA) removed from its website (2013) the list where it gave the ORAC value of every vegetable, fruit, spice and herb we consume.
 ORAC is a method for measuring antioxidant capacities in biological samples, nothing more.
ORAC is a method for measuring antioxidant capacities in biological samples, nothing more.
[
That alone is an indication of a product’s antioxidant concentration, but that’s about it.
ORAC, laboratory analysis tool
(Source: LASEVE Laboratory, Université du Québec à Chicoutimi)
ORAC is a good indication but many other aspects need to be considered. It is also necessary to know the active antioxidant molecule (or molecules) and on which human functions it acts. It is also necessary to ensure its effectiveness in penetrating cells because its cellular penetration can be low and even zero. This is the case of several drugs that must be given intravenously because, in consumable form, they do not pass through the wall of the stomach and are destroyed by the gastric process.
It is therefore a multitude of scientific analyses that are necessary to judge the quality and effectiveness of an antioxidant to intervene favorably on a function of our body. The ORAC test is therefore a laboratory analysis tool sometimes provided for in certain product research or analysis protocols but cannot alone determine the effectiveness of a product.
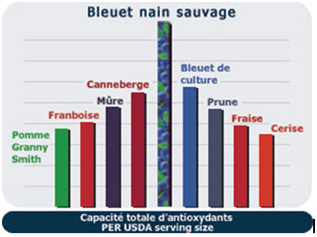
Spices and some herbs have much higher natural concentrations of antioxidants than fruits and vegetables. For example, on the ORAC scale (USDA 2002) that was publicly available, the ORAC total of cranberries was about 4 times higher than plum and twice as high as Granny Smith apple. On the other hand, on this same list and with the same scale of value, the ORAC of fresh sage was more than thirty times higher than that of cranberry and that of cloves more than 300 times.
If the antioxidant concentration of a product were directly related to health intake, a clove and an apple a day would be more than enough to live very old while keeping the doctor at bay. The only known medicinal use of cloves is the use of its flower buds to make dental analgesics as they have antiseptic and anestheticproperties. So, for cloves in particular, the relationship between ORAC and maintaining good health in general simply does not validate. This specific example clearly demonstrates that a high ORAC is not necessarily the guarantee of an effective antioxidant. It’s a promising indication but that’s it.
It should be noted that ORAC or not, of all the fruits and vegetables we consume regularly, wild blueberries are according to the USDA, at the top of the list for its total antioxidant capacity.

Finally, it is necessary to ensure that the method and scale used by manufacturers to determine the ORAC of their product is the same as that used by scientists.
11.0 Mixing does not mean adding
Some products on the market consist of mixing and potting a multitude of berry powders such as raspberries, blueberries, cherries, grapes, strawberries, blackcurrants and especially apples (given their low cost). There are similar products made with vegetables, spices and herbs. Depending on the type of fruits, vegetables, spices or herbs and the quality of the powders, some of these mixtures certainly have excellent nutritional and also medicinal qualities. No doubt about it. However, the advertising of some of these manufacturers suggests that the final blend takes all the possible benefits of each of its components while adding their ORAC values. These types of products are certainly good for health but from there to be presented as miracle products capable of intervening at the same time on all the functions of the body and thus guarantee a long, prosperous and healthy life, we are far from the account. Mixing together excellent medicinal ingredients and directing their synergy so that they can act on specific functions of the body is not an easy task. Just like with apple, it is not true that adding cloves to your blueberry juice will help protect you from cancer and also toothache.

Mixing together and consuming fruits and vegetables (fresh, frozen or in powder form) remains the best way to ensure the intake of a wide variety of excellent antioxidants. Note that some people have specific deficiencies and the consumption of certain foods, spices or herbs is recommended to them. This is the case of cranberries for people who suffer from recurrent urinary tract infections and wild blueberries for those seeking to protect themselves from cancer, cardiovascular diseases and certain degenerative diseases such as Alzheimer’s and Parkinson’s. The consumption of these berries does not cure these diseases. It can only help prevent them.
To finish with orAC, here is an example of a situation: Mixing a liter of water at 40 C. with a liter of condensed fruit juice at a temperature of 40 C. will give well 2 liters of a new mixture, but the temperature will not rise to 80 C. It is the same with the ORAC of mixed products that will not add up. In science, as soon as we change the state of matter or a variable in a formulation, we must start all the analyses from scratch. To know the result of the new mixture, the ORAC test will be one of the tools to use. However, it is to be expected that the ORAC obtained by a mixture of different fruits or vegetables will be lower than the one with the highest original ORAC.
12.0 Medicinal Properties of Berries Used in Phytonutrient Canada Products
12.1 Medicinal properties of wild dwarf blueberries
(Vaccinium angustifolium)
Wild dwarf blueberries also play an important role in the prevention of several chronic diseases, mainly due to the presence of phenolic compounds especially anthocyanins and proanthocyans (w 8-9). Wild dwarf blueberry extract (Vaccinium augustifolium) has antioxidant properties that protect cells from free radical attacks (11-25). These properties are mainly explained by the presence of powerful antioxidant compounds called anthocyanosides. Several studies in humans demonstrate that anthocyanosides (26-34) extracted from blueberries are absorbed by the body and distributed in part in blood serum (35-40). These studies also show that blueberry anthocyanosides increase the antioxidant capacity of blood serum (35-40).
The pharmacological properties attributed to blueberry extracts are numerous. Research in rats indicates that blueberry extracts have beneficial effects on the brain (41-48). They protect neurons from free radical toxicity. The authors also report that blueberries significantly improve the motor skills of older animals (42). Further work done on a mouse with Alzheimer’s shows that blueberries can prevent the onset of the disease despite the genetic predispositions of these rodents (45). Other studies conducted by this research group report that blueberries increase the memory and cognitive behavior of rats. (46-48). In addition, blueberry extracts have anticarcinogenic properties, that is, they prevent the cancerization of cells and the appearance of tumors. (49-53). They also block tumor growth by inhibiting the formation of blood vessels nourishing the tumor (angiogenesis) (53-55).
Blueberries contain a significant amount of resveratrol (56-57). This compound inhibits the aggregation of blood platelets and the oxidation of LDL (low-density lipoproteins). Resveratrol has protective effects on the cardiovascular system and helps prevent arteriosclerosis (58-59). Blueberry extract is reported to be effective for the treatment of peripheral vascular diseases, that is, it has allowed, in clinical trials, an improvement in blood circulation in pregnant women with chronic venous insufficiency. In addition, these extracts have proven useful in the pre- and postoperative treatment of varicose veins and acute phases of hemorrhoids (w 6). Marazzoni and Bombardelli (1996) demonstrated that anthocyanins have direct and indirect effects on collagen, which is the main protein element of connective tissues (tendons and cartilage). The reticular structure of anthocyanins therefore strengthens the collagen matrix. In addition, they are reported to be bioactive molecules inhibiting enzymes that break down collagen.
Another study looked at the antimicrobial property of certain fractions of wild dwarf blueberries on certain pathogenic bacteria. The results suggest that blueberry extracts are effective against two pathogenic bacteria, Listeria monocytogenesis and Escherichia coli O157:H7,better known as Listeria and E. Coli. Hippuric acid, contained in blueberry and cranberry juice is the active ingredient in the treatment of urinary tract infections caused by E. coli (w 10). The latter adheres to the wall of the bladder and urinary tract by its adhesives (designated MS and MR) located on the pili, thus allowing massive bacterial colonization. Blueberry contains a heat-stable, trypsin-resistant, non-dialysis inhibitor that acts as an anti-stickin that blocks the attachment of bacteria to the walls of the urinary tract and consequently reduces bacterial infection (w 12).
Several studies have also been conducted to evaluate the effectiveness of blueberries on type 2 diabetes. Professor Pierre Haddad Ph.D.’s team conducted a study on the effect of wild blueberries on diabetes. The researchers tested the effects of blueberry juice on a group of mice prone to obesity, diabetes and hypertension. This study made it possible to observe the potential of this juice in the fight against type 2 diabetes in humans.
Télé Québec program on the work of Dr. Pierre Haddad from the Université de Montréal:
http://lecodechastenay.telequebec.tv/occurrence.aspx?id=174&rub=4
12.4 Medicinal properties of cranberries
(Vaccinium macrocarpon)

Large-fruited cranberry is a creeping evergreen woody plant that forms carpets on the ground. The stems, thin, branched and intertwined, are 30-150 cm long. The leaves are 5 to 18 mm long (usually between 7 and 10 mm). Lingonberry(V. vitis–idaea L.) resembles cranberries but belongs to a different subgenus. The large-fruited cranberry(V. macrocarpon)is native to North America. It is found from Newfoundland and central Minnesota to Nova Scotia, New England, Long Island,New York, West Virginia, northern Ohio, central Indiana, northern Illinois, Arkansas (where the species is rare), and the Appalachian Mountains of Tennessee and North Carolina.
European settlers appreciated the value of cranberries in preventing scurvy (legitimate medicinal use given that berries are rich in vitamin C). In folk medicine, cranberries were also used to treat urinary tract infections, and this application has also been validated. It is a powerful diuretic (agent that promotes urinary excretion), and the juice is often used to treat urinary tract infections, kidney disorders and other conditions in which urinary excretion is desirable. Many women suffer from cystitis (inflammation and infection of the bladder) at some point in their lives, and it is even estimated that one in five women develop cystitis at least once a year.
Cystitis is most often caused by bacteria migrating from the urethra, and because the urethra is shorter in women, it is more easily contaminated with organisms that colonize the vagina and perineum. This is why 50% more urinary tract infections are observed in women than in men. Antibiotics are effective drugs, but they often cause side effects, can be expensive, and can lead to the development of resistant organisms.
Cranberries can be used both for preventive purposes and as an adjunct treatment for urinary tract infections. Cranberry juice treatment may require the absorption of one liter (or chopine) of pure cranberry juice per day, which few people are willing to do. Fortunately, cranberry capsules are easy to swallow.
Consumers should be aware that some « cranberry juice » found on the market may contain large amounts of sugar and added water, and that pure juice or concentrate would likely be more beneficial. In addition to preventing urinary tract infections, cranberries would deodorize the urinary tract.
Several theories have been put forward to explain why cranberries are effective in maintaining urinary tract health: cranberry juice can help acidify urine, so the bacteria that cause infection are not likely to grow in an acidic environment (this explanation no longer receives much support today); cranberry can cause the excretion in the urine of relatively large amounts of hippuric acid, which is a bacteriostatic agent; Some components present in cranberry juice can prevent bacteria from attaching to the walls of the bladder and urethra, so they are excreted during urination. It is this last explanation that is generally favored today. It seems that Escherichia coli,the bacteria most often responsible for urinary tract infections, produces constituents called adhesives that allow it to adhere to tissues, and that cranberry juice contains anti-stickines.
According to the results of preliminary trials carried out by researchers at the University of Wisconsin, cranberry juice has an antioxidant effect on clogged arteries, which would prevent cardiovascular disease.
Cranberries are extremely rich in vitamin C, moderately rich in vitamin A and have a relatively high content of fiber and anthocyanins, all components that promote good health. Hippuric acid is an important medicinal constituent of the fruit since the metabolism of this compound lowers the pH of the urine, unlike most other fruits, which, as we pointed out earlier, could explain why cranberry juice is used as an antiseptic of the urinary tract. (Compared with bear grape treatment, which is only effective as a urinary antiseptic if the urine is alkaline.)
Source 10.4: Full text parts: Government of Canada, Agriculture and Agri-Food Canada, Canadian Medicinal Crops, Vaccinium macrocarpon Ait. Full text available on the net at:
| N° RÉFÉRENCES – REFERENCES |
| (1) Boveris A, Chance B. (1973). The mitochondrial generation of hydrogen peroxide: general properties and effect of hyperbaric oxygen. Biochem. J. 134: 707-716. |
| (2) Evans P, Halliwell B. (1999). Free radicals and hearing. Cause, consequence, and criteria. Ann N Y Acad Sci. 28: 19-40. |
| (3) Halliwell B. (1991) Reactive oxygen species in living systems: source, biochemistry, and role in human disease. Am J Med. 91:14S-22S. |
| (4) Halliwell B. (1993). The role of oxygen radicals in human disease, with particular reference to the vascular system. Haemostasis. 23 Suppl 1:118–26. |
| (5) Halliwell B. (2001). Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging. 18: 685-716. |
| (6) Inoue M, Sato EF, Nishikawa M, Park AM, Kira Y, Imada I, Utsumi K. (2003). Mitochondrial generation of reactive oxygen species and its role in aerobic life. Curr Med Chem. 10: 2495-505. |
| (7) Djordjevic VB. (2004). Free radicals in cell biology. Int Rev Cytol. 237: 57-89. |
| (8) Halliwell B. (1996) Antioxidants in human health and disease. Annu Rev Nutr. 16: 33-50. |
| (9) Wickens AP. (2001). Ageing and the free radical theory. Breathing Physiology. 128: 379-391. |
| (10) Commercial Boreal Forest Research Consortium. (2000). In the realm of the boreal forest. 64 pages. |
| (11) Cao G, Shukitt-Hale B, Bickford PC, Joseph JA, McEwen J, Prior RL. (1999) Hyperoxia-induced changes in antioxidant capacity and the effect of dietary antioxidants. J Appl Physiol. 86: 1817-22. |
| (12) Kalt W, Forney CF, Martin A, Prior RL. (1999). Antioxidant capacity, vitamin C, phenolics, and anthocyanins after fresh storage of small fruits. J Agric Food Chem. 47: 4638-44. |
| (13) Wang Sy, Jiao H. (2000). Scavenging capacity of berry crops on superoxide radicals, hydrogen peroxide, hydroxyl radicals, and singlet oxygen. J Agric Food Chem. 48: 5677-84. |
| (14) Ehlenfeldt MK, Prior RL. (2001). Oxygen radical absorbance capacity (ORAC) and phenolic and anthocyanin concentrations in fruit and leaf tissues of highbush blueberry. J Agric Food Chem. 49: 2222-7. |
| (15) Connor AM, Luby JJ, Hancock JF, Berkheimer S, Hanson EJ. (2002). Changes in fruit antioxidant activity among blueberry cultivars during cold-temperature storage. J Agric Food Chem. 50: 893-8. |
| (16) Halvorsen BL, Holte K, Myhrstad MC, Barikmo I, Hvattum E, Remberg SF, Wold AB, Haffner K, Baugerod H, Andersen LF, Moskaug O, Jacobs DR Jr, Blomhoff R. (2002). A systematic screening of total antioxidants in dietary plants. J Nutr. 132: 461-71. |
| (17) Ono M, Masuoka C, Koto M, Tateishi M, Komatsu H, Kobayashi H, Igoshi K, Ito Y, Okawa M, Nohara T. (2002). Antioxidant ortho-benzoyloxyphenyl acetic acid ester, vaccihein A, from the fruit of rabbiteye blueberry (Vaccinium ashei). Chem Pharm Bull (Tokyo). 50: 1416-7. |
| (18) Zheng W, Wang SY. Oxygen radical absorbing capacity of phenolics in blueberries, cranberries, chokeberries, and lingonberries. (2003). J Agric Food Chem. 51: 502-9. |
| (19) Zheng Y, Wang Cy, Wang SY, Zheng W. (2003). Effect of high-oxygen atmospheres on blueberry phenolics, anthocyanins, and antioxidant capacity. J Agric Food Chem. 51: 7162-9. |
| (20) Ichikawa H, Ichiyanagi T, Xu B, Yoshii Y, Nakajima M, Konishi T. (2001). Antioxidant Activity of Anthocyanin Extract from Purple Black Rice. J Med Food. 4: 211-218. |
| (21) Prior RL, Hoang H, Gu L, Wu X, Bacchiocca M, Howard L, Hampsch-Woodill M, Huang D, Ou B, Jacob R. (2003). Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORAC(FL))) of plasma and other biological and food samples. J Agric Food Chem. 51: 3273-9. |
| (22) Cevallos-Casals BA, Cisneros-Zevallos L. (2003). Stoichiometric and kinetic studies of phenolic antioxidants from Andean purple corn and red-fleshed sweetpotato. J Agric Food Chem. 51: 3313-9. |
| (23) Blomhoff R. Antioxidants and oxidative stress. (2004). Tidsskr Nor Laegeforen. 124: 1643-5. |
| (24) Lichtenthaler R, Marx F. (2005). Total oxidant scavenging capacities of common European fruit and vegetable juices. J Agric Food Chem. 53: 103-10. |
| (25) Parry J, Su L, Luther M, Zhou K, Yurawecz MP, Whittaker P, Yu L. (2005). Fatty acid composition and antioxidant properties of cold-pressed marionberry, boysenberry, red raspberry, and blueberry seed oils. J Agric Food Chem. 53: 566-73. |
| (26) Ichiyanagi T, Tateyama C, Oikawa K, Konishi T. (2000). Comparison of anthocyanin distribution in different blueberry sources by capillary zone electrophoresis. Biol Pharm Bull. 23: 492-7. |
| (27) Ichiyanagi T, Oikawa K, Tateyama C, Konishi T. (2001). Acid mediated hydrolysis of blueberry anthocyanins. Chem Pharm Bull (Tokyo). 49: 114-7. |
| (28) Kalt W, Ryan DA, Duy JC, Prior RL, Ehlenfeldt MK, Vander Kloet SP. (2001). Interspecific variation in anthocyanins, phenolics, and antioxidant capacity among genotypes of highbush and lowbush blueberries (Vaccinium section cyanococcus spp.). J Agric Food Chem. 49: 4761-7. |
| (29) Sellappan S, Akoh CC, Krewer G. Phenolic compounds and antioxidant capacity of Georgia-grown blueberries and blackberries. (2002). J Agric Food Chem. 50: 2432-8. |
| (30) Gu L, Kelm M, Hammerstone JF, Beecher G, Cunningham D, Vannozzi S, Prior RL. (2002). Fractionation of polymeric procyanidins from lowbush blueberry and quantification of procyanidins in selected foods with an optimized normal-phase HPLC-MS fluorescent detection method. J Agric Food Chem. 50: 4852-60. |
| (31) Sanchez-Moreno C, Cao G, Or B, Prior RL. (2003). Anthocyanin and proanthocyanidin content in selected white and red wines. Oxygen radical absorbance capacity comparison with nontraditional wines obtained from highbush blueberry. J Agric Food Chem. 51: 4889-96. |
| (32) Taruscio TG, Barney DL, Exon J. (2004). Content and profile of flavanoid and phenolic acid compounds in conjunction with the antioxidant capacity for a variety of northwest Vaccinium berries. J Agric Food Chem. 52: 3169-76. |
| (33) Schmidt BM, Howell AB, McEniry B, Knight CT, Rye D, Erdman JW Jr, Lila MA. (2004). Effective separation of potent antiproliferation and antiadhesion components from wild blueberry (Vaccinium angustifolium Ait.) fruits. J Agric Food Chem. 52: 6433-42. |
| (34) Nakajima JI, Tanaka I, Seo S, Yamazaki M, Saito K. (2004). LC/PDA/ESI-MS Profiling and Radical Scavenging Activity of Anthocyanins in Various Berries. J Biomed Biotechnol. 2004: 241-247. |
| (35) Pedersen CB, Kyle J, Jenkinson AM, Gardner PT, McPhail DB, Duthie GG. (2000). Effects of blueberry and cranberry juice consumption on the plasma antioxidant capacity of healthy female volunteers. Eur J Clin Nutr. 54: 405-8. |
| (36) Wu X, Cao G, Prior RL. (2002). Absorption and metabolism of anthocyanins in elderly women after consumption of elderberry or blueberry. J Nutr. 132: 1865-71. |
| (37) Kay CD, Holub BJ. (2002). The effect of wild blueberry (Vaccinium angustifolium) consumption on postprandial serum antioxidant status in human subjects. Br J Nutr. 88: 389-98. |
| (38) Mazza G, Kay CD, Cottrell T, Holub BJ. (2002). Absorption of anthocyanins from blueberries and serum antioxidant status in human subjects. J Agric Food Chem. 50: 7731-7. |
| (39) Kay CD, Mazza G, Holub BJ, Wang J. (2004). Anthocyanin metabolites in human urine and serum. Br J Nutr. 91: 933-42. |
| (40) Ichiyanagi T, Rahman MM, Kashiwada Y, Ikeshiro Y, Shida Y, Hatano Y, Matsumoto H, Hirayama M, Tsuda T, Konishi T. (2004). Absorption and metabolism of delphinidin 3-O-beta-D-glucopyranoside in rats. Free Radic Biol Med. 36: 930-7. |
| (41) Joseph JA, Denisova N, Fisher D, Shukitt-Hale B, Bickford P, Prior R, Cao G. (1998). Membrane and receptor modifications of oxidative stress vulnerability in aging. Nutritional considerations. Ann N Y Acad Sci. 854: 268-76. |
| (42) Joseph JA, Shukitt-Hale B, Denisova NA, Bielinski D, Martin A, McEwen JJ, Bickford PC. (1999). Reversals of age-related declines in neuronal signal transduction, cognitive, and motor behavioral deficits with blueberry, spinach, or strawberry dietary supplementation. J Neurosci. 19: 8114-21. |
| (43) Joseph JA, Denisova NA, Bielinski D, Fisher DR, Shukitt-Hale B. (2000). Oxidative stress protection and vulnerability in aging: putative nutritional implications for intervention. Mech Ageing Dev. 116: 141-53. |
| (44) Galli RL, Shukitt-Hale B, Youdim KA, Joseph JA. (2002). Fruit polyphenolics and brain aging: nutritional interventions targeting age-related neuronal and behavioral deficits. Ann N Y Acad Sci. 959: 128-32. |
| (45) Joseph JA, Denisova NA, Arendash G, Gordon M, Diamond D, Shukitt-Hale B, Morgan D. (2003). Blueberry supplementation enhances signaling and prevents behavioral deficits in an Alzheimer disease model. Nutr Neurosci. 6: 153-62. |
| (46) Goyarzu P, Malin DH, Lau FC, Taglialatela G, Moon WD, Jennings R, Moy E, Moy D, Lippold S, Shukitt-Hale B, Joseph JA. (2004). Blueberry supplemented diet: effects on object recognition memory and nuclear factor-kappa B levels in aged rats. Nutr Neurosci. 7: 75-83. |
| (47) Joseph JA, Shukitt-Hale B, Casadesus G. (2005). Reversing the deleterious effects of aging on neuronal communication and behavior: beneficial properties of fruit polyphenolic compounds. Am J Clin Nutr. 81(1 Suppl): 313S-316S. |
| (48) Casadesus G, Shukitt-Hale B, Stellwagen HM, Zhu X, Lee HG, Smith MA, Joseph JA. (2004). Modulation of hippocampal plasticity and cognitive behavior by short-term blueberry supplementation in aged rats. Nutr Neurosci. 7: 309-16. |
| (49) Bomser J, Madhavi DL, Singletary K, Smith MA. (1996). In vitro anticancer activity of fruit extracts from Vaccinium species. Planta Med. 62: 212-6. |
| (50) Kapadia GJ, Azuine MA, Tokuda H, Hang E, Mukainaka T, Nishino H, Sridhar R. (2002). Inhibitory effect of herbal remedies on 12-O-tetradecanoylphorbol-13-acetate-promoted Epstein-Barr virus early antigen activation. Pharmacol Res. 45: 213-20. |
| (51) Wedge DE, Meepagala KM, Magee JB, Smith SH, Huang G, Larcom LL. (2001). Anticarcinogenic Activity of Strawberry, Blueberry, and Raspberry Extracts to Breast and Cervical Cancer Cells. J Med Food. 4: 49-51. |
| (52) Hope Smith S, Tate PL, Huang G, Magee JB, Meepagala KM, Wedge DE, Larcom LL. (2004). Antimutagenic activity of berry extracts. J Med Food. 7: 450-5. |
| (53) Bagchi D, Sen CK, Bagchi M, Atalay M. (2004). Anti-angiogenic, antioxidant, and anti-carcinogenic properties of a novel anthocyanin-rich berry extract formula. Biochemistry (Mosc). 69: 75-80, 1 p preceding 75. |
| (54) Roy S, Khanna S, Alessio HM, Vider J, Bagchi D, Bagchi M, Sen CK. (2002). Anti-angiogenic property of edible berries. Free Radic Res. 36: 1023-31. |
| (55) Atalay M, Gordillo G, Roy S, Rovin B, Bagchi D, Bagchi M, Sen CK. (2003). Anti-angiogenic property of edible berry in a model of hemangioma. FEBS Lett. 544: 252-7. |
| (56) Lyons MM, Yu C, Toma RB, Cho SY, Reiboldt W, Lee J, van Breemen RB. (2003). Resveratrol in raw and baked blueberries and bilberries. J Agric Food Chem. 51: 5867-70. |
| (57) Rimando AM, Kalt W, Magee JB, Dewey J, Ballington JR. (2004). Resveratrol, pterostilbene, and piceatannol in vaccinium berries. J Agric Food Chem. 52: 4713-9. |
| (58) Fremont L. (2000). Biological effects of resveratrol. Life Sci. 66: 663-73. |
| (59) Wu JM, Wang ZR, Hsieh TC, Bruder JL, Zou JG, Huang YZ. (2001). Mechanism of cardioprotection by resveratrol, a phenolic antioxidant present in red wine (Review). Int J Mol Med. 8: 3-17. |
| (60) Brother Marie-Victorin. (1964). Laurentian Flora. The presses of the University of Montreal. 2nd edition. P.439. |
| (61) Ek S, Kartimo H, Mattila S, Tolonen A. (2006). Characterization of phenolic compounds from lingonberry (Vaccinium vitis-idaea). J Agric Food Chem. 54: 9834-42. |
| (62) Wang SY, Feng R, Bowman L, Penhallegon R, Ding M, Lu Y. (2005) Antioxidant activity in lingonberries (Vaccinium vitis-idaea L.) and its inhibitory effect on activator protein-1, nuclear factor-kappaB, and mitogen-activated protein kinases activation. J Agric Food Chem. 53: 3156-66. |
| (63) Viljanen K, Kylli P, Hubbermann EM, Schwarz K, Heinonen M. (2005). Anthocyanin antioxidant activity and partition behavior in whey protein emulsion. J Agric Food Chem. 53: 2022-7. |
| (64) Lichtenthaler R, Marx F. (2005) Total oxidant scavenging capacities of common European fruit and vegetable juices. J Agric Food Chem. 53: 103-10. |
| (65) Viljanen K, Kylli P, Kivikari R, Heinonen M. (2004). Inhibition of protein and lipid oxidation in liposomes by berry phenolics. J Agric Food Chem. 52: 7419-24. |
| (66) Zheng W, Wang SY. (2003). Oxygen radical absorbing capacity of phenolics in blueberries, cranberries, chokeberries, and lingonberries. J Agric Food Chem. 51: 502-9. |
| (67) Ho KY, Huang JS, Tsai CC, Lin TC, Hsu YF, Lin CC (1999). Antioxidant activity of tannin components from Vaccinium vitis-idaea L. J Pharm Pharmacol. 51: 1075-8. |
| (68) Ho KY, Tsai CC, Huang JS, Chen CP, Lin TC, Lin CC. Antimicrobial activity of tannin components from Vaccinium vitis-idaea L. (2001). J Pharm Pharmacol. 53: 187-91. |
| (69) Gotsman I, Lotan C, Soskolne WA, Rassovsky S, Pugatsch T, Lapidus L, Novikov Y, Masrawa S, Stabholz A. (2007). Periodontal Destruction Is Associated With Coronary Artery Disease and Periodontal Infection With Acute Coronary Syndrome. J Periodontol. 78: 849-858. |
| (70) Nohynek LJ, Alakomi HL, Kahkonen MP, Heinonen M, Helander IM, Oksman-Caldentey KM, Puupponen-Pimia RH. (2006) Berry phenolics: antimicrobial properties and mechanisms of action against severe human pathogens. Nutr Cancer. 54:18-32. |
| (71) Puupponen-Pimia R, Nohynek L, Hartmann-Schmidlin S, Kahkonen M, Heinonen M, Maatta-Riihinen K, Oksman-Caldentey KM. (2005). Berry phenolics selectively inhibit the growth of intestinal pathogens. J Appl Microbiol. 98: 991-1000. |
| (72) Tunon H, Olavsdotter C, Bohlin L. (1995). Evaluation of anti-inflammatory activity of some Swedish medicinal plants. Inhibition of prostaglandin biosynthesis and PAF-induced exocytosis. J Ethnopharmacol. 48: 61-76. |
| (73) Paivarinta E, Pajari AM, Torronen R, Mutanen M. (2006). Ellagic acid and natural sources of ellagitannins as possible chemopreventive agents against intestinal tumorigenesis in the Min mouse. Nutr Cancer. 54:79-83. |
| (74) Aggarwal BB, Shishodia S. (2006). Molecular targets of dietary agents for prevention and therapy of cancer. Biochem Pharmacol. 71:1397-421. |
| (75) Barta I, Smerak P, Polivkova Z, Sestakova H, Langova M, Turek B, Bartova J. (2006) Current trends and perspectives in nutrition and cancer prevention. Neoplasma. 53:19-25. |
| (76) Huetz P, Mavaddat N, Mavri J. (2005). Reaction between ellagic acid and an ultimate carcinogen. J Chem Inf Model. 45: 1564-70. |
| (77) Pavlica S, Gebhardt R. (2005). Protective effects of ellagic and chlorogenic acids against oxidative stress in PC12 cells. Free Radic Res. 39: 1377-90. |
| (78) Dorai T, Aggarwal BB. (2004). Role of chemopreventive agents in cancer therapy. Cancer Lett. 215: 129-40. |
| (79) Festa F, Aglitti T, Duranti G, Ricordy R, Perticone P, Cozzi R. (2001). Strong antioxidant activity of ellagic acid in mammalian cells in vitro revealed by the comet assay. Anticancer Res. 21: 3903-8. |
| (80) Wargovich MJ. (1997). Experimental evidence for cancer preventive elements in foods. Cancer Lett. 114: 11-7. |
| (81) Stoner GD, Mukhtar H. Polyphenols as cancer chemopreventive agents. (1995). J Cell Biochem Suppl. 22:169-80. |
| (82) Szarka CE, Grana G, Engstrom PF. (1994). Chemoprevention of cancer. Curr Probl Cancer. 18: 6-79. |
| (83) Kelloff GJ, Boone CW, Crowell JA, Steele VE, Lubet R, Sigman CC. (1994) Chemopreventive drug development: perspectives and progress. Cancer Epidemiol Biomarkers Prev. 3: 85-98. |
| (84) Hayatsu H, Arimoto S, Negishi T. (1988). Dietary inhibitors of mutagenesis and carcinogenesis. Mutat Res. 202:429-46. |
| (85) Tidsskr Nor Laegeforen. 2003 Jun 26;123(13-14):1856-7. [Cloudberry: an important source of ellagic acid, an anti-oxidant].[Article in Norwegian] Thiem B, Berge V. Spring Department of Pharmaceutical Botany, K. Marcinkowski University of Medical Sciences, Pozńa, Poland. Abstract Cloudberry, Rubus chamaemorus L. (Rosaceae) is an herbaceous plant with a boreal, circumpolar distribution. Its fruit is of economic importance in northern Scandinavia; furthermore, this plant is of significance in Norwegian tradition. We present a review of its biology, secondary metabolites as well as cloudberry applications. The compounds of primary interest in cloudberry are vitamin C and ellagotannins with a high level of ellagic acid that exhibits biological activities. Ellagic acid, a dietary phenolic, offers considerable promise as anticarcinogen, antimutagen and antioxidant. The ellagic acid content in cloudberries could have health benefits. |
| (86) J Agric Food Chem. 2011 Apr 13;59(7):3330-9. Epub 2011 Mar 4.Food grade lingonberry extract: polyphenolic composition and in vivo protective effect against oxidative stress. Mane C, Loonis M, Juhel C, Dufour C, Malian-Aubert C. Spring
CHR-Hansen-SAS, 92 avenue des Baronnes, 34730 Prades-le-Lez, France. Abstract Fractionation of the polyphenols constituting a food grade lingonberry extract (Vaccinium vitis-idaea) highlighted a composition more complex than described until now in the berry. Procyanidins B1, B2, and A2 were identified by UPLC/ESI-MS(2) along with the presence of other flavanol oligomers. Processing induced the release of large amounts of aglycones for ferulic acid, p-coumaric acid, and quercetin. The described anthocyanic composition of lingonberry was completed with hexoside derivatives of peonidin, petunidin, malvidin, and delphinidin. Besides confirmation of in vitro antioxidant activity, in vivo study was performed on rats fed a diet inducing oxidative stress. Supplementation with lingonberry extract significantly decreased the total oxidant status and favorably affected antioxidant defense enzymes in red blood cells and liver. A drop in the serum reduced glutathione level was also prevented, and uric acid was maintained at low level, confirming the antioxidant activity of the extract (5% proanthocyanidins) from a dosage of 23 mg/kg of body weight. |
| N° RÉFÉRENCES – REFERENCES | |
| (1) Boveris A, Chance B. (1973). The mitochondrial generation of hydrogen peroxide: general properties and effect of hyperbaric oxygen. Biochem. J. 134: 707-716. | |
| (2) Evans P, Halliwell B. (1999). Free radicals and hearing. Cause, consequence, and criteria. Ann N Y Acad Sci. 28: 19-40. | |
| (3) Halliwell B. (1991) Reactive oxygen species in living systems: source, biochemistry, and role in human disease. Am J Med. 91:14S-22S. | |
| (4) Halliwell B. (1993). The role of oxygen radicals in human disease, with particular reference to the vascular system. Haemostasis. 23 Suppl 1:118–26. | |
| (5) Halliwell B. (2001). Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging. 18: 685-716. | |
| (6) Inoue M, Sato EF, Nishikawa M, Park AM, Kira Y, Imada I, Utsumi K. (2003). Mitochondrial generation of reactive oxygen species and its role in aerobic life. Curr Med Chem. 10: 2495-505. | |
| (7) Djordjevic VB. (2004). Free radicals in cell biology. Int Rev Cytol. 237: 57-89. | |
| (8) Halliwell B. (1996) Antioxidants in human health and disease. Annu Rev Nutr. 16: 33-50. | |
| (9) Wickens AP. (2001). Ageing and the free radical theory. Breathing Physiology. 128: 379-391. | |
| (10) Commercial Boreal Forest Research Consortium. (2000). In the realm of the boreal forest. 64 pages. | |
| (11) Cao G, Shukitt-Hale B, Bickford PC, Joseph JA, McEwen J, Prior RL. (1999) Hyperoxia-induced changes in antioxidant capacity and the effect of dietary antioxidants. J Appl Physiol. 86: 1817-22. | |
| (12) Kalt W, Forney CF, Martin A, Prior RL. (1999). Antioxidant capacity, vitamin C, phenolics, and anthocyanins after fresh storage of small fruits. J Agric Food Chem. 47: 4638-44. | |
| (13) Wang Sy, Jiao H. (2000). Scavenging capacity of berry crops on superoxide radicals, hydrogen peroxide, hydroxyl radicals, and singlet oxygen. J Agric Food Chem. 48: 5677-84. | |
| (14) Ehlenfeldt MK, Prior RL. (2001). Oxygen radical absorbance capacity (ORAC) and phenolic and anthocyanin concentrations in fruit and leaf tissues of highbush blueberry. J Agric Food Chem. 49: 2222-7. | |
| (15) Connor AM, Luby JJ, Hancock JF, Berkheimer S, Hanson EJ. (2002). Changes in fruit antioxidant activity among blueberry cultivars during cold-temperature storage. J Agric Food Chem. 50: 893-8. | |
| (16) Halvorsen BL, Holte K, Myhrstad MC, Barikmo I, Hvattum E, Remberg SF, Wold AB, Haffner K, Baugerod H, Andersen LF, Moskaug O, Jacobs DR Jr, Blomhoff R. (2002). A systematic screening of total antioxidants in dietary plants. J Nutr. 132: 461-71. | |
| (17) Ono M, Masuoka C, Koto M, Tateishi M, Komatsu H, Kobayashi H, Igoshi K, Ito Y, Okawa M, Nohara T. (2002). Antioxidant ortho-benzoyloxyphenyl acetic acid ester, vaccihein A, from the fruit of rabbiteye blueberry (Vaccinium ashei). Chem Pharm Bull (Tokyo). 50: 1416-7. | |
| (18) Zheng W, Wang SY. Oxygen radical absorbing capacity of phenolics in blueberries, cranberries, chokeberries, and lingonberries. (2003). J Agric Food Chem. 51: 502-9. | |
| (19) Zheng Y, Wang Cy, Wang SY, Zheng W. (2003). Effect of high-oxygen atmospheres on blueberry phenolics, anthocyanins, and antioxidant capacity. J Agric Food Chem. 51: 7162-9. | |
| (20) Ichikawa H, Ichiyanagi T, Xu B, Yoshii Y, Nakajima M, Konishi T. (2001). Antioxidant Activity of Anthocyanin Extract from Purple Black Rice. J Med Food. 4: 211-218. | |
| (21) Prior RL, Hoang H, Gu L, Wu X, Bacchiocca M, Howard L, Hampsch-Woodill M, Huang D, Ou B, Jacob R. (2003). Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORAC(FL))) of plasma and other biological and food samples. J Agric Food Chem. 51: 3273-9. | |
| (22) Cevallos-Casals BA, Cisneros-Zevallos L. (2003). Stoichiometric and kinetic studies of phenolic antioxidants from Andean purple corn and red-fleshed sweetpotato. J Agric Food Chem. 51: 3313-9. | |
| (23) Blomhoff R. Antioxidants and oxidative stress. (2004). Tidsskr Nor Laegeforen. 124: 1643-5. | |
| (24) Lichtenthaler R, Marx F. (2005). Total oxidant scavenging capacities of common European fruit and vegetable juices. J Agric Food Chem. 53: 103-10. | |
| (25) Parry J, Su L, Luther M, Zhou K, Yurawecz MP, Whittaker P, Yu L. (2005). Fatty acid composition and antioxidant properties of cold-pressed marionberry, boysenberry, red raspberry, and blueberry seed oils. J Agric Food Chem. 53: 566-73. | |
| (26) Ichiyanagi T, Tateyama C, Oikawa K, Konishi T. (2000). Comparison of anthocyanin distribution in different blueberry sources by capillary zone electrophoresis. Biol Pharm Bull. 23: 492-7. | |
| (27) Ichiyanagi T, Oikawa K, Tateyama C, Konishi T. (2001). Acid mediated hydrolysis of blueberry anthocyanins. Chem Pharm Bull (Tokyo). 49: 114-7. | |
| (28) Kalt W, Ryan DA, Duy JC, Prior RL, Ehlenfeldt MK, Vander Kloet SP. (2001). Interspecific variation in anthocyanins, phenolics, and antioxidant capacity among genotypes of highbush and lowbush blueberries (Vaccinium section cyanococcus spp.). J Agric Food Chem. 49: 4761-7. | |
| (29) Sellappan S, Akoh CC, Krewer G. Phenolic compounds and antioxidant capacity of Georgia-grown blueberries and blackberries. (2002). J Agric Food Chem. 50: 2432-8. | |
| (30) Gu L, Kelm M, Hammerstone JF, Beecher G, Cunningham D, Vannozzi S, Prior RL. (2002). Fractionation of polymeric procyanidins from lowbush blueberry and quantification of procyanidins in selected foods with an optimized normal-phase HPLC-MS fluorescent detection method. J Agric Food Chem. 50: 4852-60. | |
| (31) Sanchez-Moreno C, Cao G, Or B, Prior RL. (2003). Anthocyanin and proanthocyanidin content in selected white and red wines. Oxygen radical absorbance capacity comparison with nontraditional wines obtained from highbush blueberry. J Agric Food Chem. 51: 4889-96. | |
| (32) Taruscio TG, Barney DL, Exon J. (2004). Content and profile of flavanoid and phenolic acid compounds in conjunction with the antioxidant capacity for a variety of northwest Vaccinium berries. J Agric Food Chem. 52: 3169-76. | |
| (33) Schmidt BM, Howell AB, McEniry B, Knight CT, Rye D, Erdman JW Jr, Lila MA. (2004). Effective separation of potent antiproliferation and antiadhesion components from wild blueberry (Vaccinium angustifolium Ait.) fruits. J Agric Food Chem. 52: 6433-42. | |
| (34) Nakajima JI, Tanaka I, Seo S, Yamazaki M, Saito K. (2004). LC/PDA/ESI-MS Profiling and Radical Scavenging Activity of Anthocyanins in Various Berries. J Biomed Biotechnol. 2004: 241-247. | |
| (35) Pedersen CB, Kyle J, Jenkinson AM, Gardner PT, McPhail DB, Duthie GG. (2000). Effects of blueberry and cranberry juice consumption on the plasma antioxidant capacity of healthy female volunteers. Eur J Clin Nutr. 54: 405-8. |
|
| (36) Wu X, Cao G, Prior RL. (2002). Absorption and metabolism of anthocyanins in elderly women after consumption of elderberry or blueberry. J Nutr. 132: 1865-71. | |
| (37) Kay CD, Holub BJ. (2002). The effect of wild blueberry (Vaccinium angustifolium) consumption on postprandial serum antioxidant status in human subjects. Br J Nutr. 88: 389-98. | |
| (38) Mazza G, Kay CD, Cottrell T, Holub BJ. (2002). Absorption of anthocyanins from blueberries and serum antioxidant status in human subjects. J Agric Food Chem. 50: 7731-7. | |
| (39) Kay CD, Mazza G, Holub BJ, Wang J. (2004). Anthocyanin metabolites in human urine and serum. Br J Nutr. 91: 933-42. | |
| (40) Ichiyanagi T, Rahman MM, Kashiwada Y, Ikeshiro Y, Shida Y, Hatano Y, Matsumoto H, Hirayama M, Tsuda T, Konishi T. (2004). Absorption and metabolism of delphinidin 3-O-beta-D-glucopyranoside in rats. Free Radic Biol Med. 36: 930-7. | |
| (41) Joseph JA, Denisova N, Fisher D, Shukitt-Hale B, Bickford P, Prior R, Cao G. (1998). Membrane and receptor modifications of oxidative stress vulnerability in aging. Nutritional considerations. Ann N Y Acad Sci. 854: 268-76. | |
| (42) Joseph JA, Shukitt-Hale B, Denisova NA, Bielinski D, Martin A, McEwen JJ, Bickford PC. (1999). Reversals of age-related declines in neuronal signal transduction, cognitive, and motor behavioral deficits with blueberry, spinach, or strawberry dietary supplementation. J Neurosci. 19: 8114-21. | |
| (43) Joseph JA, Denisova NA, Bielinski D, Fisher DR, Shukitt-Hale B. (2000). Oxidative stress protection and vulnerability in aging: putative nutritional implications for intervention. Mech Ageing Dev. 116: 141-53. | |
| (44) Galli RL, Shukitt-Hale B, Youdim KA, Joseph JA. (2002). Fruit polyphenolics and brain aging: nutritional interventions targeting age-related neuronal and behavioral deficits. Ann N Y Acad Sci. 959: 128-32. | |
| (45) Joseph JA, Denisova NA, Arendash G, Gordon M, Diamond D, Shukitt-Hale B, Morgan D. (2003). Blueberry supplementation enhances signaling and prevents behavioral deficits in an Alzheimer disease model. Nutr Neurosci. 6: 153-62. | |
| (46) Goyarzu P, Malin DH, Lau FC, Taglialatela G, Moon WD, Jennings R, Moy E, Moy D, Lippold S, Shukitt-Hale B, Joseph JA. (2004). Blueberry supplemented diet: effects on object recognition memory and nuclear factor-kappa B levels in aged rats. Nutr Neurosci. 7: 75-83. | |
| (47) Joseph JA, Shukitt-Hale B, Casadesus G. (2005). Reversing the deleterious effects of aging on neuronal communication and behavior: beneficial properties of fruit polyphenolic compounds. Am J Clin Nutr. 81(1 Suppl): 313S-316S. | |
| (48) Casadesus G, Shukitt-Hale B, Stellwagen HM, Zhu X, Lee HG, Smith MA, Joseph JA. (2004). Modulation of hippocampal plasticity and cognitive behavior by short-term blueberry supplementation in aged rats. Nutr Neurosci. 7: 309-16. | |
| (49) Bomser J, Madhavi DL, Singletary K, Smith MA. (1996). In vitro anticancer activity of fruit extracts from Vaccinium species. Planta Med. 62: 212-6. | |
| (50) Kapadia GJ, Azuine MA, Tokuda H, Hang E, Mukainaka T, Nishino H, Sridhar R. (2002). Inhibitory effect of herbal remedies on 12-O-tetradecanoylphorbol-13-acetate-promoted Epstein-Barr virus early antigen activation. Pharmacol Res. 45: 213-20. | |
| (51) Wedge DE, Meepagala KM, Magee JB, Smith SH, Huang G, Larcom LL. (2001). Anticarcinogenic Activity of Strawberry, Blueberry, and Raspberry Extracts to Breast and Cervical Cancer Cells. J Med Food. 4: 49-51. | |
| (52) Hope Smith S, Tate PL, Huang G, Magee JB, Meepagala KM, Wedge DE, Larcom LL. (2004). Antimutagenic activity of berry extracts. J Med Food. 7: 450-5. | |
| (53) Bagchi D, Sen CK, Bagchi M, Atalay M. (2004). Anti-angiogenic, antioxidant, and anti-carcinogenic properties of a novel anthocyanin-rich berry extract formula. Biochemistry (Mosc). 69: 75-80, 1 p preceding 75. | |
| (54) Roy S, Khanna S, Alessio HM, Vider J, Bagchi D, Bagchi M, Sen CK. (2002). Anti-angiogenic property of edible berries. Free Radic Res. 36: 1023-31. | |
| (55) Atalay M, Gordillo G, Roy S, Rovin B, Bagchi D, Bagchi M, Sen CK. (2003). Anti-angiogenic property of edible berry in a model of hemangioma. FEBS Lett. 544: 252-7. | |
| (56) Lyons MM, Yu C, Toma RB, Cho SY, Reiboldt W, Lee J, van Breemen RB. (2003). Resveratrol in raw and baked blueberries and bilberries. J Agric Food Chem. 51: 5867-70. | |
| (57) Rimando AM, Kalt W, Magee JB, Dewey J, Ballington JR. (2004). Resveratrol, pterostilbene, and piceatannol in vaccinium berries. J Agric Food Chem. 52: 4713-9. | |
| (58) Fremont L. (2000). Biological effects of resveratrol. Life Sci. 66: 663-73. | |
| (59) Wu JM, Wang ZR, Hsieh TC, Bruder JL, Zou JG, Huang YZ. (2001). Mechanism of cardioprotection by resveratrol, a phenolic antioxidant present in red wine (Review). Int J Mol Med. 8: 3-17. | |
| (60) Brother Marie-Victorin. (1964). Laurentian Flora. The presses of the University of Montreal. 2nd edition. P.439. | |
| (61) Ek S, Kartimo H, Mattila S, Tolonen A. (2006). Characterization of phenolic compounds from lingonberry (Vaccinium vitis-idaea). J Agric Food Chem. 54: 9834-42. | |
| (62) Wang SY, Feng R, Bowman L, Penhallegon R, Ding M, Lu Y. (2005) Antioxidant activity in lingonberries (Vaccinium vitis-idaea L.) and its inhibitory effect on activator protein-1, nuclear factor-kappaB, and mitogen-activated protein kinases activation. J Agric Food Chem. 53: 3156-66. | |
| (63) Viljanen K, Kylli P, Hubbermann EM, Schwarz K, Heinonen M. (2005). Anthocyanin antioxidant activity and partition behavior in whey protein emulsion. J Agric Food Chem. 53: 2022-7. | |
| (64) Lichtenthaler R, Marx F. (2005) Total oxidant scavenging capacities of common European fruit and vegetable juices. J Agric Food Chem. 53: 103-10. | |
| (65) Viljanen K, Kylli P, Kivikari R, Heinonen M. (2004). Inhibition of protein and lipid oxidation in liposomes by berry phenolics. J Agric Food Chem. 52: 7419-24. | |
| (66) Zheng W, Wang SY. (2003). Oxygen radical absorbing capacity of phenolics in blueberries, cranberries, chokeberries, and lingonberries. J Agric Food Chem. 51: 502-9. | |
| (67) Ho KY, Huang JS, Tsai CC, Lin TC, Hsu YF, Lin CC (1999). Antioxidant activity of tannin components from Vaccinium vitis-idaea L. J Pharm Pharmacol. 51: 1075-8. | |
| (68) Ho KY, Tsai CC, Huang JS, Chen CP, Lin TC, Lin CC. Antimicrobial activity of tannin components from Vaccinium vitis-idaea L. (2001). J Pharm Pharmacol. 53: 187-91. | |
| (69) Gotsman I, Lotan C, Soskolne WA, Rassovsky S, Pugatsch T, Lapidus L, Novikov Y, Masrawa S, Stabholz A. (2007). Periodontal Destruction Is Associated With Coronary Artery Disease and Periodontal Infection With Acute Coronary Syndrome. J Periodontol. 78: 849-858. | |
| (70) Nohynek LJ, Alakomi HL, Kahkonen MP, Heinonen M, Helander IM, Oksman-Caldentey KM, Puupponen-Pimia RH. (2006) Berry phenolics: antimicrobial properties and mechanisms of action against severe human pathogens. Nutr Cancer. 54:18-32. | |
| (71) Puupponen-Pimia R, Nohynek L, Hartmann-Schmidlin S, Kahkonen M, Heinonen M, Maatta-Riihinen K, Oksman-Caldentey KM. (2005). Berry phenolics selectively inhibit the growth of intestinal pathogens. J Appl Microbiol. 98: 991-1000. | |
| (72) Tunon H, Olavsdotter C, Bohlin L. (1995). Evaluation of anti-inflammatory activity of some Swedish medicinal plants. Inhibition of prostaglandin biosynthesis and PAF-induced exocytosis. J Ethnopharmacol. 48: 61-76. | |
| (73) Paivarinta E, Pajari AM, Torronen R, Mutanen M. (2006). Ellagic acid and natural sources of ellagitannins as possible chemopreventive agents against intestinal tumorigenesis in the Min mouse. Nutr Cancer. 54:79-83. | |
| (74) Aggarwal BB, Shishodia S. (2006). Molecular targets of dietary agents for prevention and therapy of cancer. Biochem Pharmacol. 71:1397-421. | |
| (75) Barta I, Smerak P, Polivkova Z, Sestakova H, Langova M, Turek B, Bartova J. (2006) Current trends and perspectives in nutrition and cancer prevention. Neoplasma. 53:19-25. | |
| (76) Huetz P, Mavaddat N, Mavri J. (2005). Reaction between ellagic acid and an ultimate carcinogen. J Chem Inf Model. 45: 1564-70. | |
| (77) Pavlica S, Gebhardt R. (2005). Protective effects of ellagic and chlorogenic acids against oxidative stress in PC12 cells. Free Radic Res. 39: 1377-90. | |
| (78) Dorai T, Aggarwal BB. (2004). Role of chemopreventive agents in cancer therapy. Cancer Lett. 215: 129-40. | |
| (79) Festa F, Aglitti T, Duranti G, Ricordy R, Perticone P, Cozzi R. (2001). Strong antioxidant activity of ellagic acid in mammalian cells in vitro revealed by the comet assay. Anticancer Res. 21: 3903-8. | |
| (80) Wargovich MJ. (1997). Experimental evidence for cancer preventive elements in foods. Cancer Lett. 114: 11-7. | |
| (81) Stoner GD, Mukhtar H. Polyphenols as cancer chemopreventive agents. (1995). J Cell Biochem Suppl. 22:169-80. | |
| (82) Szarka CE, Grana G, Engstrom PF. (1994). Chemoprevention of cancer. Curr Probl Cancer. 18: 6-79. | |
| (83) Kelloff GJ, Boone CW, Crowell JA, Steele VE, Lubet R, Sigman CC. (1994) Chemopreventive drug development: perspectives and progress. Cancer Epidemiol Biomarkers Prev. 3: 85-98. | |
| (84) Hayatsu H, Arimoto S, Negishi T. (1988). Dietary inhibitors of mutagenesis and carcinogenesis. Mutat Res. 202:429-46. | |
| (85) Tidsskr Nor Laegeforen. 2003 Jun 26;123(13-14):1856-7. [Cloudberry: an important source of ellagic acid, an anti-oxidant].[Article in Norwegian] Thiem B, Berge V. Spring Department of Pharmaceutical Botany, K. Marcinkowski University of Medical Sciences, Pozńa, Poland. Abstract Cloudberry, Rubus chamaemorus L. (Rosaceae) is an herbaceous plant with a boreal, circumpolar distribution. Its fruit is of economic importance in northern Scandinavia; furthermore, this plant is of significance in Norwegian tradition. We present a review of its biology, secondary metabolites as well as cloudberry applications. The compounds of primary interest in cloudberry are vitamin C and ellagotannins with a high level of ellagic acid that exhibits biological activities. Ellagic acid, a dietary phenolic, offers considerable promise as anticarcinogen, antimutagen and antioxidant. The ellagic acid content in cloudberries could have health benefits. |
|
| (86) J Agric Food Chem. 2011 Apr 13;59(7):3330-9. Epub 2011 Mar 4.Food grade lingonberry extract: polyphenolic composition and in vivo protective effect against oxidative stress. Mane C, Loonis M, Juhel C, Dufour C, Malian-Aubert C. Spring
CHR-Hansen-SAS, 92 avenue des Baronnes, 34730 Prades-le-Lez, France. Abstract Fractionation of the polyphenols constituting a food grade lingonberry extract (Vaccinium vitis-idaea) highlighted a composition more complex than described until now in the berry. Procyanidins B1, B2, and A2 were identified by UPLC/ESI-MS(2) along with the presence of other flavanol oligomers. Processing induced the release of large amounts of aglycones for ferulic acid, p-coumaric acid, and quercetin. The described anthocyanic composition of lingonberry was completed with hexoside derivatives of peonidin, petunidin, malvidin, and delphinidin. Besides confirmation of in vitro antioxidant activity, in vivo study was performed on rats fed a diet inducing oxidative stress. Supplementation with lingonberry extract significantly decreased the total oxidant status and favorably affected antioxidant defense enzymes in red blood cells and liver. A drop in the serum reduced glutathione level was also prevented, and uric acid was maintained at low level, confirming the antioxidant activity of the extract (5% proanthocyanidins) from a dosage of 23 mg/kg of body weight. |
|



